Physiology Friday #312: An Inconsistent Bedtime Doubles Cardiovascular Disease Risk
The stability of when you sleep might be as important as how long you're in bed.
Greetings!
Welcome to the Physiology Friday newsletter.
Details about the sponsors of this newsletter and deals on products I love, including Ketone-IQ, Create creatine, Equip Foods, and ProBio Nutrition can be found at the end of the post.
Today, I’m discussing what I think is probably one of the most important studies on sleep in the last decade.
It suggests that the timing and consistency of your sleep (especially when you go to bed) may be just as important as how long you sleep.
Sleep health research has been handicapped because we’ve been looking at a question that’s much too simple: how much sleep do people get?
That question has produced an enormous literature linking both short (and long) sleep duration with cardiometabolic disease, mortality, impaired glucose regulation, hypertension, and obesity… among other maladies. “The shorter your sleep, the shorter your life.”
But duration increasingly looks like only one dimension of sleep health. A person can sleep for an apparently adequate number of hours while still living in a state of circadian misalignment—going to bed at widely varying times, waking at inconsistent hours, and forcing their physiology to adapt to shifting time schedules.
The emerging idea is that sleep may not merely be a matter of quantity or even quality, but also of consistency and regularity… whether the timing of sleep is stably embedded within your body’s natural 24-hour cycle.
Cardiovascular physiology is profoundly circadian. Blood pressure, endothelial function, hormone secretion, glucose handling, and even our susceptibility to heart attacks (they’re more common in the morning) all fluctuate across the day-night cycle. When behavior repeatedly misaligns with those oscillations—as in shift work, late chronotypes (“night owls”), or erratic sleep timing—the result is far worse than just poor sleep. For systems like the cardiovascular system that depend on some level of “biological predictability,” this may spell disaster for long-term health.
In fact, the American Heart Association has recently updated its stance on sleep to reflect this emerging data.1 Their recent scientific statement marks a clear shift from treating sleep mainly as a duration issue to treating it as a multidimensional health behavior, with sleep regularity elevated to a core component of cardiometabolic health. The AHA explicitly frames healthy sleep as including not just duration, but also continuity, timing, regularity, daytime functioning, sleep architecture, and the absence of sleep disorders. Regularity is now being emphasized alongside quantity and quality as something that may shape blood pressure, glucose regulation, adiposity, and other cardiometabolic pathways.
The authors of a new study position sleep timing regularity within exactly this framework… as a measurable expression of circadian stability. And they find that regularity matters, a LOT. Especially if you’re not sleeping enough.2
The study, from the Northern Finland Birth Cohort 1966, asks a more specific question than much of the existing sleep-regularity literature. Rather than relying only on composite (and frankly difficult to understand) metrics such as the Sleep Regularity Index or interdaily stability (which are inherently hard to calculate and even harder to implement practically), it isolates three highly interpretable timing variables:
Bedtime
Wake-up time
The midpoint of the sleep period
A standard deviation in bedtime or wake-up time is immediately legible as a behavioral pattern. If risk concentrates there, the biology becomes easier to interpret, and the intervention target becomes more concrete. And that’s one of the reasons this study is so knowledgeable and practical.
The cohort included 3,231 adults, all around midlife, drawn from participants in the 46-year follow-up of the study conducted in 2012–2014. These individuals wore a wrist-based activity monitor continuously for up to two weeks, and sleep timing variables were derived using a sleep-detection algorithm that’s been validated against both self-reported sleep diaries and smart-ring-derived sleep periods. In other words, it’s pretty accurate at assessing sleep.
The authors used the first seven consecutive days of valid monitoring, reasoning that a seven-day window captures both weekday and weekend structure and is sufficient to estimate someone’s habitual timing variability.
Bedtime, wake-up time, and sleep midpoint were each quantified as the standard deviation across those seven days—the main outcome being sleep timing regularity. Participants were categorized into one of three sleep timing regularity categories:
Regular
Fairly regular
Irregular
To get a better idea of how “regular” or “irregular” these people’s sleep patterns were, let’s look at the median values for sleep timing across each of these groups. In other words, how much did bedtime, wake-up time, and the midpoint of sleep vary from night to night?
For regular sleepers, bedtime had a variability of 33 minutes, wake-up time had a variability of 41 minutes, and the midpoint of sleep had a variability of 33 minutes.
For fairly regular sleepers, variability for bedtime, wake-up time, and sleep midpoint was 59 minutes, 73 minutes, and 55 minutes, respectively.
For the irregular sleepers, variability was 108 minutes, 114 minutes, and 93 minutes for bedtime, wake-up time, and sleep midpoint, respectively.
Let’s put this in perspective. This means that someone with an irregular sleep schedule might have a bedtime or wake-up time that changes by 1.5 to 2 hours from night to night. They might usually go to bed at 11 pm, but might also go to bed at 9 pm on some nights and as late as 1 am on others. But someone with a regular schedule and an average bedtime of 11 pm will be in bed between 10:30 and 11:30 every single night.
The primary endpoint of this study was major adverse cardiac events (obtained from a national health registry), defined as the first time someone experienced acute myocardial infarction, unstable angina, stroke, hospitalization for heart failure, or cardiovascular death.
The authors also divided participants based on the median sleep duration of the group—7 hours 56 minutes—creating below-median and above-median sleeper groups (let’s just refer to them as “short sleepers” and “normal sleepers.”
During a follow-up period of about 130 months, 128 participants—4.0% of the sample—experienced a major cardiovascular event. But surprisingly, those who had an event did not differ significantly from those who didn’t in average bedtime, average wake-up time, sleep midpoint, or how many hours they slept each night. On the surface, then, there was no simple signal that people who later developed cardiovascular events were just sleeping later, waking later, or sleeping less overall.
And this is where the paper becomes interesting…
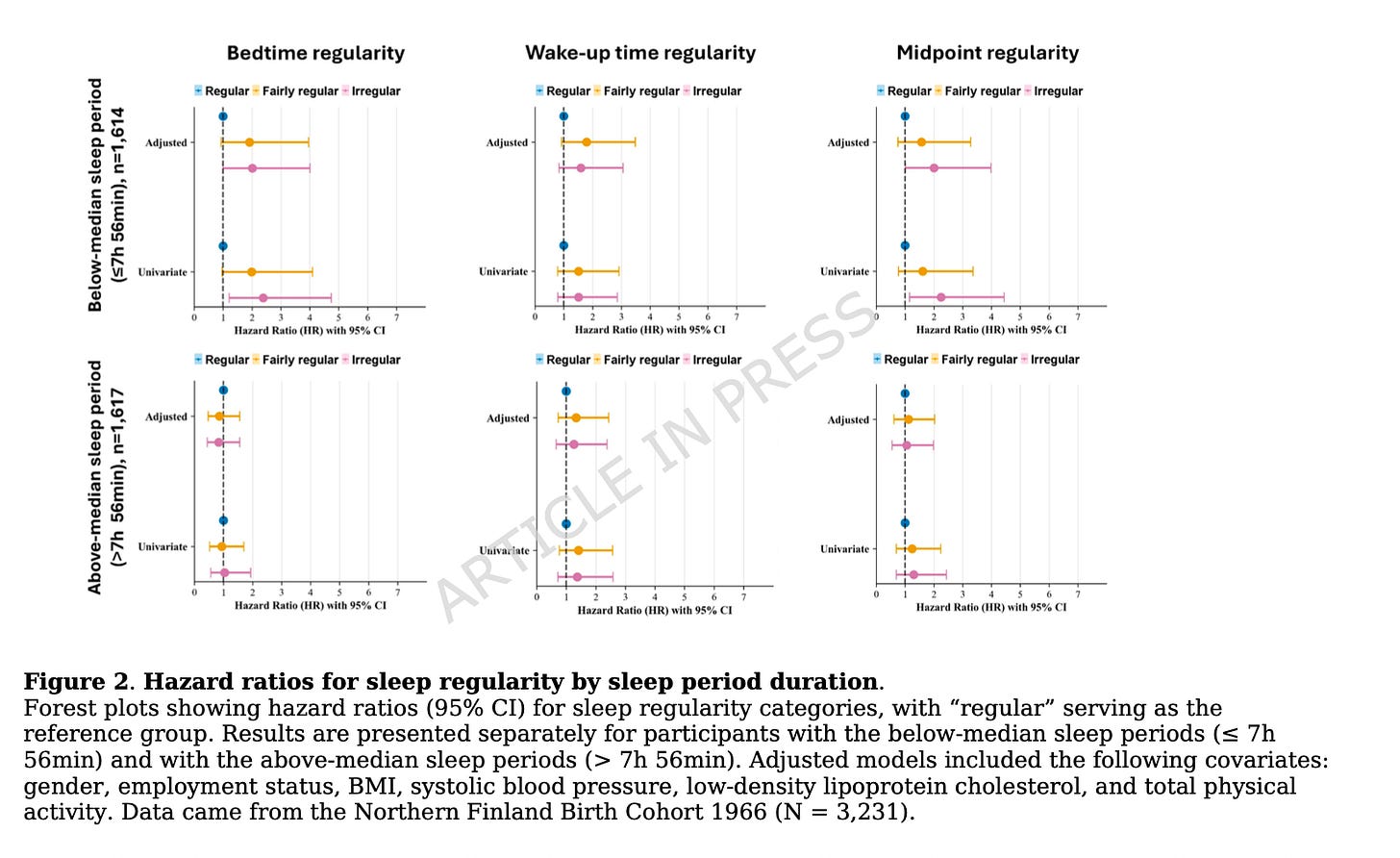
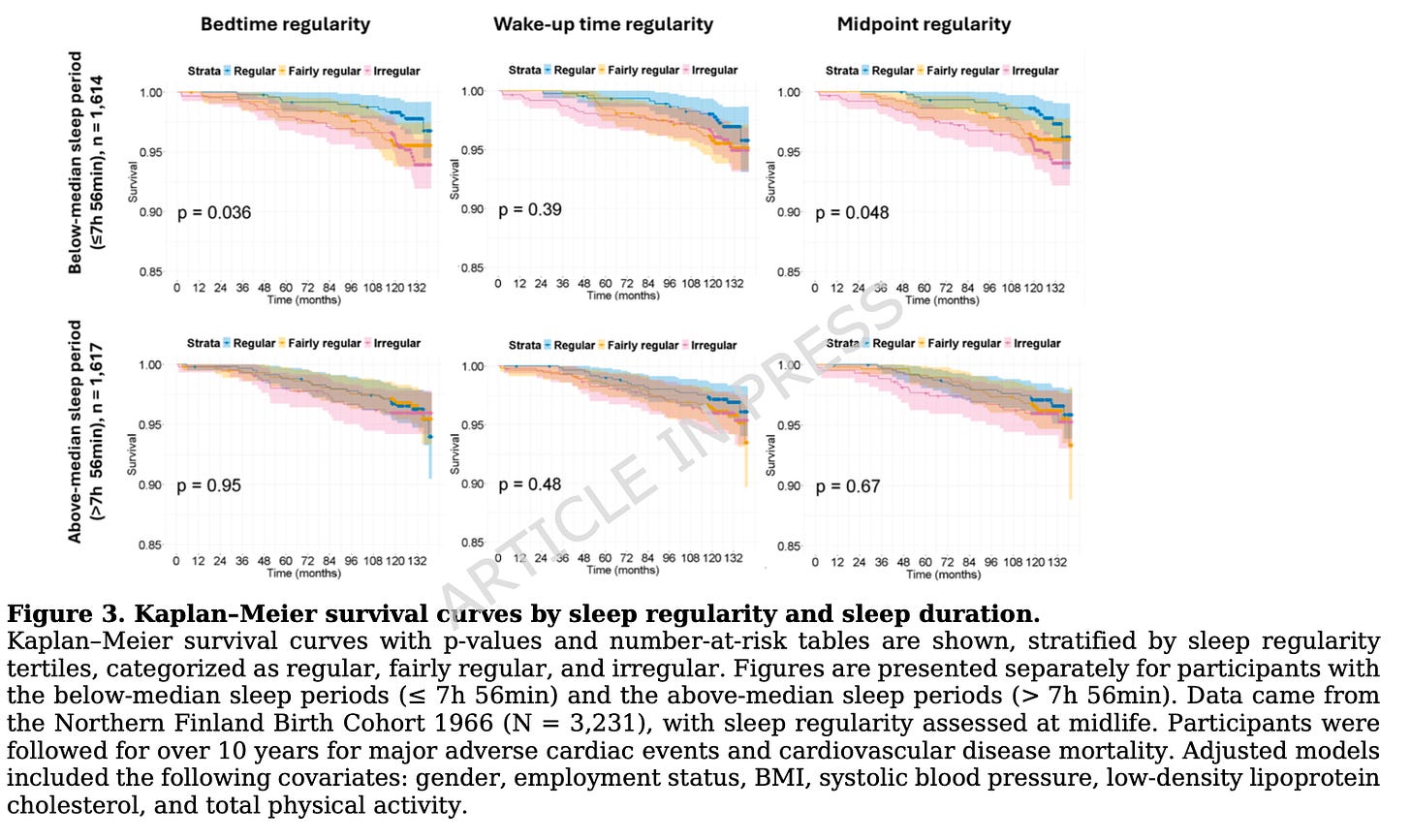
Among participants whose sleep period duration fell at or below the median—7 hours 56 minutes or less—irregular sleep timing predicted a higher cardiovascular risk.
Compared with those in the most regular bedtime category, participants with irregular bedtimes had twice the risk of experiencing a major cardiovascular event during the follow-up.
Participants with irregular sleep midpoints also had a 2-fold greater risk of a major cardiovascular event compared to regular sleepers.
Wake-up time irregularity was not significantly associated with risk.
Importantly, none of the three timing variables predicted cardiovascular events among participants with “normal sleep”—those getting more than 7 hours and 56 minutes of sleep per night!
This is a very particular result, and it somewhat contradicts previous findings that sleep regularity is harmful even among people getting enough sleep. This study wouldn’t support that. It’s only among people sleeping somewhat less than eight hours that the instability of when one goes to bed (and their midpoint of sleep) seems to matter for heart disease risk.
A few more important nuances. Even when the researchers excluded the 12 participants who experienced a major cardiovascular event within the first two years of follow-up, the results did not materially change, which argues against the simplest version of reverse causation (the idea that subclinical cardiovascular disease at baseline was already disrupting sleep timing and driving the association).
Excluding shift workers also did not change the results. In fact, the associations became slightly stronger when shift workers (a group with notoriously altered circadian rhythms) were excluded. After removing shift workers, participants with irregular bedtimes and sleep midpoints had a 2.25–2.3x greater risk of cardiovascular events during the follow-up.
That matters because it suggests irregular sleep timing in ordinary life—outside classic occupational circadian disruption—may itself mark a meaningful health hazard. It wasn’t the shift workers in the study “artificially” driving the association.
One of the more intriguing (and to me, surprising) features of the study is the interaction, or at least the modification, of sleep duration. The association appears only in those with “short sleep.” This can be read in two ways.
One interpretation is that irregular timing becomes especially harmful when it is layered onto relatively shorter sleep, because the combination compounds insufficient recovery and circadian disruption.
Another is that longer sleep may buffer some of the physiological consequences of timing irregularity. The authors lean toward the latter possibility in their own conclusion, suggesting that sufficient sleep may offer partial protection against an irregular bedtime.
And I think this matters for how you should think about your own sleep. If you aren’t getting enough sleep each night, then your strategy needs to be to anchor when you go to bed as close to the same time as possible every night. But if you’re getting enough sleep, you have more “wiggle room.”
The study suggests that not all components of sleep regularity are equivalent. It may be the consistency of initiating sleep—or the consistency of the overall phase of the sleep episode—rather than the consistency of waking per se, that carries more relevance to cardiovascular health. Why might that be?
One plausible explanation is that bedtime is the “entry point” into the nighttime restorative window, whereas wake-up time is often a more downstream, socially constrained event. Sleep onset is tightly coupled to circadian phase. People typically fall asleep as melatonin is rising, core body temperature is falling, and the circadian system is shifting toward the nighttime state.
If bedtime is pushed around from night to night, that phase relationship becomes unstable, so the person is not just sleeping at different clock times but repeatedly asking the cardiovascular, endocrine, and autonomic systems to enter sleep from different circadian states (different “body times”).
A second reason bedtime may matter more is that it likely has a greater influence over sleep architecture and early-night recovery biology. Slow-wave sleep and slow-wave activity are concentrated in the early part of the night, whereas REM sleep predominates later in the night. So when bedtime shifts later or becomes erratic, the body may repeatedly alter the alignment between the first part of sleep and the conditions that normally favor restorative processes. Wake-up time variability, in contrast, may mostly affect the tail end of sleep and may be less “biologically informative” if many people still wake at roughly similar times because of work or family obligations. In that sense, bedtime irregularity may be a better marker of chronic circadian misalignment and of disrupted access to the most important portion of sleep.
We have to note one of the main limitations here. Sleep timing regularity was measured over only seven days at baseline, which may or may not capture an individual’s longer-term habitual behavior across seasons, stressors, or life transitions. How someone slept during one particular week might not reflect how they slept during the nearly 11-year follow-up period.
What this paper ultimately does is suggest that cardiovascular sleep epidemiology (and our own lifestyle practices) may need to move beyond how long people sleep and even beyond when they usually sleep, toward how stably sleep is placed from one day to the next. Regularity and consistency should become a focus of sleep health equal to that of sleep duration.
For now, this study does not argue that sleep regularity matters more than every other cardiovascular risk factor, but it does show that the stability of sleep—specifically bedtime and sleep midpoint—predicts future major cardiovascular events in a way that average sleep timing does not.
I think this may prove to be one of the most consequential conceptual shifts in modern sleep health—from thinking of sleep as a quantity, to understanding it as a rhythm.
Thanks for reading. See you next Friday.
~Brady~
Ketone-IQ is high-performance energy in a bottle. I use it for post-exercise recovery along with enhancing focus, mood, and cognition. Take 30% off your order.
Create is the first “modern creatine” brand. They sell a wide range of creatine monohydrate gummies—and yeah, their gummies actually contain creatine, unlike some other brands. They’re giving my audience 20% off their order.
ProBio Nutrition—the all-in-one supplement that I use every single day—is offering 20% off. My preference is the tangy orange flavor, but they also sell an unflavored “smoothie booster” that’s great in a shake, smoothie, or juice.
Equip Foods makes some of the cleanest, best-tasting protein products around. I am absolutely obsessed with their Prime grass-fed protein bars (the peanut butter ones are to die for). Take 35% off a subscription.








Another good argument for ending the twice annual time change so many of us are forced to endure.
Anecdotally I find even a 2 hour time shift when I visit family and return makes me feel run down.