Physiology Friday #313: A “Molecular Brake” on Fitness Explains Why Some People Don’t Respond to Exercise
Have scientists discovered the trainability gene?
Greetings!
Welcome to the Physiology Friday newsletter.
Details about the sponsors of this newsletter and deals on products I love, including Ketone-IQ, Create creatine, Equip Foods, and ProBio Nutrition can be found at the end of the post.
One of the oldest questions in exercise science is why some people seem to improve dramatically with training while others improve only modestly or, at least on paper, not at all.
That debate sits at the center of the long-running idea of “non-responders” to exercise. It’s compelling, but also contested.
This concept comes from studies showing that when a group of people completes the same training program, some increase their cardiorespiratory fitness (their VO2 max improves) while others show little change. In some cases, a few participants even appear to get worse… but that apparent decline is often more likely to reflect measurement error, day-to-day biological variability, or statistical noise than a true drop in fitness.
That’s one reason the whole idea of a true “non-responder” remains hotly debated. And I’m skeptical of it myself.
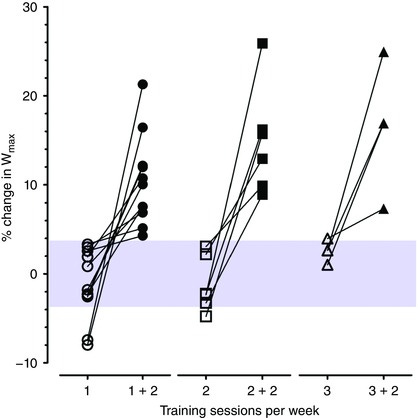
A lot of research shows that when so-called non-responders are given a larger dose of exercise (more weekly volume, a higher frequency of sessions, or more intensity), they often do respond. Their cardiorespiratory fitness improves. In other words, they may not be biologically incapable of adapting; they may simply need a different stimulus.
Probably one of my favorite exercise science studies ever was one that challenged the whole idea of “non-responders” to training. In the study, researchers took 78 healthy adults and had them complete the same endurance training program, but with different weekly doses ranging from 60 to 300 minutes per week. The more people exercised, the fewer of them were classified as non-responders.
The people initially labeled as non-responders then went on to do another 6 weeks of training, this time with an extra 120 minutes of exercise per week. Once the dose increased, they responded… non-response essentially disappeared. To me, that is such an important finding because it suggests that many so-called non-responders are probably not true non-responders at all. They may just be people who need a larger training stimulus to cross whatever threshold is required for adaptation. That idea makes a lot more physiological sense to me than the idea that exercise just somehow doesn’t “work” for a meaningful subset of people.
Putting this study aside, the idea that we all respond differently to the same training stimulus makes sense. Human physiology is variable. Biology is variable. It would be surprising if every person responded the same way to the exact same training plan. So the real question is not whether responses differ—they clearly do—but why.
What explains why one person responds robustly while another barely budges? Are there underlying biological differences driving that variability? Could genetics play a role? And are there measurable molecular or physiological markers that could help predict who is likely to respond to a given exercise program?
That is the premise of a new study pointing to a potential biomarker in the blood: a protein called insulin-like growth factor binding protein 7, or IGFBP7. In humans, higher levels predicted a worse response to training and were even associated with a range of cardiometabolic diseases. And in mice, deleting this “molecular brake” produced an enhanced training response.1
Let’s get into it.
The study comes from a multi-institutional group that included researchers from Massachusetts General Hospital, Harvard, the University of Michigan, and the Norwegian University of Science and Technology. Their work builds on the Generation 100 trial in Norway, which was a randomized clinical trial involving 1,567 older adults with an average age of 73 years.
The researchers focused on the trial’s high-intensity interval training (HIIT) arm. Participants performed two supervised HIIT sessions per week for a full year, with each session consisting of four 4-minute intervals at 90% of peak heart rate, separated by 3-minute active recovery periods (it’s the classic Norwegian 4x4 interval workout).
The variation in the responses to HIIT was enormous. After a year of identical training, changes in peak VO2 ranged from a 56% increase to a 28% decline.
That naturally raises a deeper question and goes back to the idea of exercise non-responders we talked about earlier. What is going on biologically in the people who fail to adapt?
IGFBP1 is a “molecular brake” on fitness
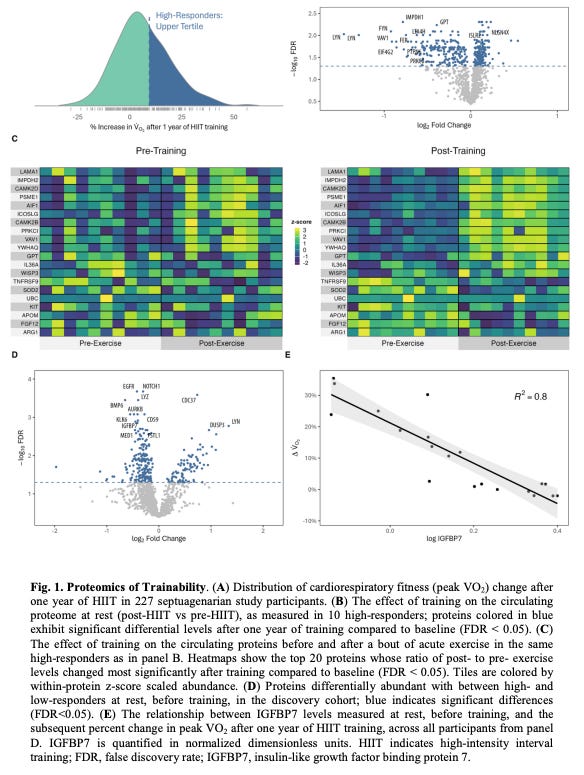
To figure out what might be blocking adaptation, the researchers moved from whole-body performance to the blood. They selected the extremes—10 high responders, who improved peak VO2 by an average of 22%, and 10 low responders, who showed no gain or a decline. Crucially, they matched them carefully before training. Same age. Same sex. Same baseline fitness. Same lean mass. This ensured that the difference between these people was not apparent in their outward phenotype.
The team measured 305 proteins under four conditions:
At rest before training,
Immediately after acute maximal exercise before training,
At rest after the year-long training intervention, and
Immediately after acute maximal exercise after training.
Now we can understand how the proteome responded to acute stress and how it changed over time after long-term training.
One of the first really important findings was that low responders were not failing to register the workout. Acute exercise produced major proteomic shifts in both groups. In fact, 659 proteins changed in high responders, while 783 proteins changed in low responders. So the people who ultimately failed to improve were not “responding less” to exercise. Their bodies absolutely detected the stress, and if anything, the acute perturbation was even larger.
After 12 months of HIIT, the high responders showed 394 significant changes in their resting proteome. Their biological baseline had changed. The low responders, by contrast, showed zero significant changes in their resting proteome. Zero. I think that is one of the most striking findings in the paper. Both groups repeatedly experienced acute stress and mounted an immediate response to exercise. But only one group appeared to ingrain this into their physiology and actually “remodel” their resting proteome.
The next step was to ask whether future responsiveness could be predicted before training even started. Using the baseline blood samples alone, the researchers identified 228 proteins that were associated with later fitness gains. And among all of these candidate proteins, one stood out most clearly: IGFBP7, or insulin-like growth factor binding protein 7.
IGFBP7 showed a striking inverse relationship with training response. Higher baseline levels predicted worse gains in fitness, and lower baseline levels predicted better gains. Baseline IGFBP7 accounted for about 80% of the variance in trainability among the participants. That is an enormous signal.
This gets even more interesting (okay, at least to me) when we consider what IGFBP7 actually is. The protein is classified as a senescence-associated secreted protein, or SASP factor, which means it is part of the inflammatory and suppressive cocktail released by senescent cells—the so-called “zombie cells” that accumulate with age. That provides a direct biological bridge between aging and blunted adaptation to exercise.
Of course, a strong correlation does not prove causation. But the problem is, without genetic engineering (which presents its own ethical problems and isn’t yet acceptable for most human research), we can’t really establish causation in humans.
So the authors turned to mouse models and started manipulating the system directly. They used IGFBP7 knockout mice, animals genetically engineered so they could not produce the protein at all. Interestingly, these mice had 14–15% less muscle mass in the heart and leg muscles, and the female knockout mice had 61% more fat mass than controls (who were able to produce IGFBP7). Yet despite these differences in body composition, the knockout mice had similar baseline exercise capacity to normal mice (just like the human study).
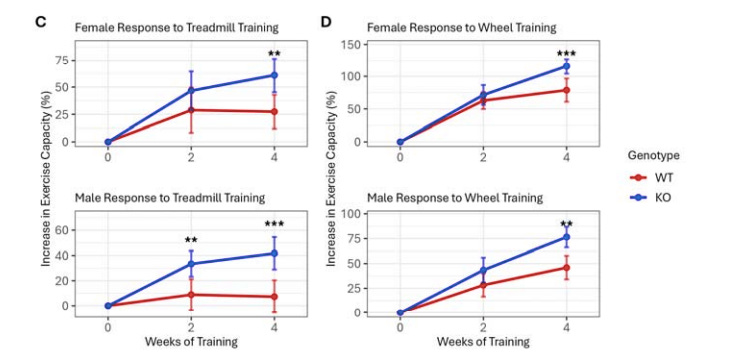
Once the mice started training, the difference became obvious. In a treadmill training protocol, the knockout mice improved dramatically more than wild-type controls. After four weeks, they gained 3.3 times more fitness overall. Male knockouts gained 5.7 times more.
The researchers then tested whether this advantage would hold up in a different kind of exercise setting. Instead of forced treadmill work, they gave the mice voluntary running wheels. Over four weeks, the animals ran 5–7 kilometers per day, which for a mouse is a huge training volume. Both groups improved more than they did on the treadmill, but the knockout mice still outperformed the wild-types by 54%. By week two, the knockouts had already pulled away. So the absence of IGFBP7 raised both the ceiling of adaptation and accelerated the kinetics, with mice adapting faster and to a greater extent when they lacked IGFBP7.
But there is still an obvious caveat with knockout models: if you remove a gene starting from conception, maybe the animal develops differently in all kinds of ways unrelated to the protein’s immediate role in adulthood. The researchers addressed that too. They increased circulating IGFBP7 in adult mice by about fourfold. Even though the manipulated mice still ran the same distances on the wheel (exercise behavior didn’t change), the enhanced trainability disappeared. This suggests IGFBP7 is not merely a biomarker of poor adaptation. It appears to actively constrain it.
So what is this protein actually doing?
IGFBP7 seems to act like a systems-level brake on several core components of exercise adaptation. One major axis is muscle growth and repair.
The paper points to evidence that IGFBP7 interferes with insulin-like growth factor-1 (IGF-1) signaling, essentially blocking one of the body’s main anabolic pathways. In the knockout mice, circulating IGF-1 was 40% higher, and the animals gained 9% more muscle mass across multiple muscles during training.
Another major axis is blood vessel growth. Trained knockout mice had about double the increase in capillary density compared with controls.
The study also found broad metabolic adaptations across the muscle, heart, and liver. With training, the knockout mice appeared to shift into a more advanced fuel-handling strategy, exhibiting greater fatty acid metabolism in skeletal muscle, more Krebs cycle and amino acid breakdown in the heart, and enhanced aerobic and fat metabolism in the liver.
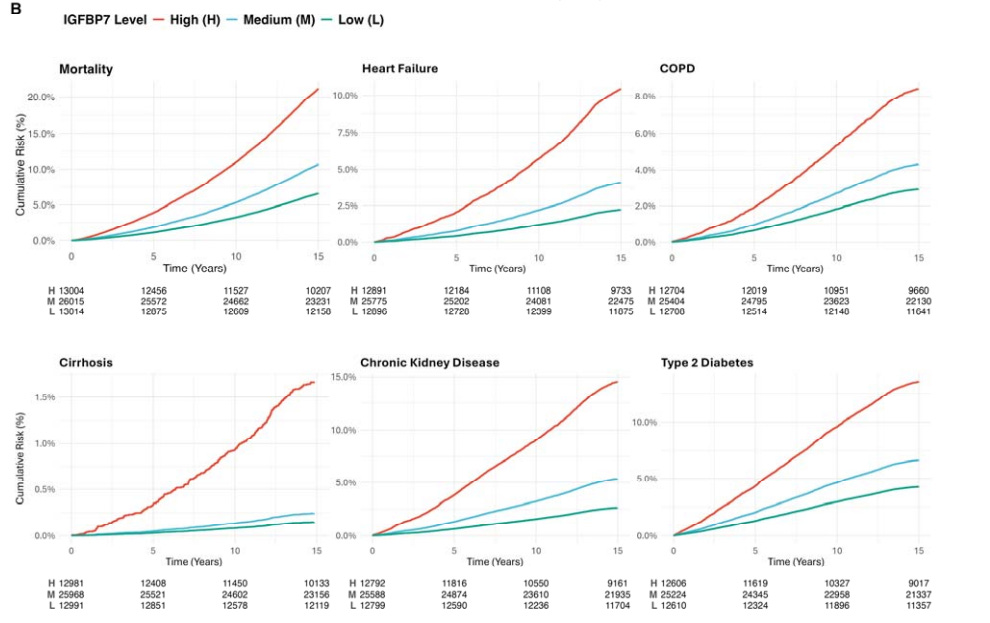
The final layer of the paper zooms out from exercise performance to long-term health (something we are all interested in) using UK Biobank data from 52,033 participants followed for about 16 years. After adjusting for age and sex, lower IGFBP7 levels were associated with a lower risk of all-cause mortality, heart failure, COPD, cirrhosis, chronic kidney disease, and type 2 diabetes. When participants were grouped based on their baseline levels of IGFBP7, those with the highest levels had a 2.9- to 11-fold greater disease rate than those with the lowest levels across these outcomes.
There’s a bit of a philosophical conclusion to this paper that I really dig.
The authors propose that trainability may reflect something deeper than exercise response per se. It may be a visible readout of cellular resilience. A hard workout is, at the cellular level, a controlled crisis. Body temperature rises, blood acidity shifts, energy demand spikes, and blood flow stress changes rapidly. Those same kinds of disturbances also show up in disease states.
Maybe the biology that allows a person to adapt to exercise is not separate from the biology that allows them to withstand chronic disease. Maybe the ability to remodel in response to training is a visible expression of a broader capacity to survive and adapt to physiological stress. If that is true, then poor trainability is both a sports-performance problem and a window into aging biology itself.
The same molecular systems that determine whether your body responds to a training block may also help determine how well it resists heart failure, kidney disease, diabetes, or the slow erosion of function that comes with age. And IGFBP7 may be one of the clearest molecular signals yet identified that helps explain why. In fact, this could be one potential explanation for the broader link between VO2 max and longevity that I’ve covered so extensively. While I do think that aerobic fitness is protective in the sense that it reflects the body’s overall capacity to coordinate oxygen delivery and energy production, this paper would suggest it may also reflect physiological plasticity—the processes that allow someone to obtain a high level of fitness in the first place are also important for guarding against death and disease.
This does not mean the obvious next step is to block IGFBP7 and call it a day. The therapeutic angle is much trickier than that. Because IGFBP7 is tied to senescence and growth control, lowering it could come with serious tradeoffs (prior work has shown that knockout mice can develop certain types of cancer). So the same brake that may limit adaptation could also exist for a reason (biology isn’t so dumb after all). It may prevent a system from becoming too permissive. The paper raises the possibility that transient or context-specific inhibition could one day help older or frail individuals get more biological return from lower-intensity activity, but that is still a speculative therapeutic vision.
Still, even setting aside the pharmaceutical angle, this is a fascinating piece of science. It gives us a candidate molecular explanation for one of the most persistent mysteries in exercise physiology. It links age-related senescence biology to adaptation and shows that the body can fully register the stress of exercise and yet still fail to remodel. And it suggests that trainability may be less about motivation or willpower than about whether someone (or some mouse) is biochemically capable of upgrading itself in response to stress.
More than anything, this paper makes me think about aging a little differently.
We often think of aging as the slow accumulation of damage. But studies like this hint that maybe aging is also a progressive loss of adaptive capacity. If that is true, then the future of healthy aging may depend on preserving the body’s ability to adapt in the first place. And this study suggests IGFBP7 might be one of the proteins sitting directly in the middle of that story.
Thanks for reading. See you next Friday.
~Brady~
Ketone-IQ is high-performance energy in a bottle. I use it for post-exercise recovery along with enhancing focus, mood, and cognition. Take 30% off your order.
Create is the first “modern creatine” brand. They sell a wide range of creatine monohydrate gummies—and yeah, their gummies actually contain creatine, unlike some other brands. They’re giving my audience 20% off their order.
ProBio Nutrition—the all-in-one supplement that I use every single day—is offering 20% off. My preference is the tangy orange flavor, but they also sell an unflavored “smoothie booster” that’s great in a shake, smoothie, or juice.
Equip Foods makes some of the cleanest, best-tasting protein products around. I am absolutely obsessed with their Prime grass-fed protein bars (the peanut butter ones are to die for). Take 35% off a subscription.